With an urgent need to standardize the interpretation of PD-L1 in Triple Negative Breast Cancer (TNBC), UK NEQAS ICC & ISH has taken the lead by offering an interpretative External Quality Assessment (EQA) module assessing pathologist’s ability to correctly score a range of clinical representative samples. The module is hosted on the PathoGate.net platform, which has been set up by the UK NEQAS team and a panel of external expert pathologist. The experts have carefully selected a small sample set, which is being assessed by participating international pathologists.


With the rapidly growing list of indications and scoring methods for PD-L1 it is important as well as challenging to ensure that the interpretation of this predictive bio-marker is accurate and precise. Many clinical labs are taking on PD-L1 testing in TNBC with limited access to training and resulting uncertainty regarding their assessment performance.
In order to address this, UK NEQAS ICC & ISH has initiated an interpretative EQA module for PD-L1 in TNBC, in which participants around the world are requested to assess a range of samples selected in collaboration with a panel of expert pathologists.
The module is hosted on the PathoGate.net platform, which allows pathologists to assess digitally scanned images of PD-L1 stained slides as well as corresponding H&E sections side by side. Participants are requested to assess the PD-L1 status of the slide (positive vs. negative) as well as a quantitative estimate of the percentage of intra-tumoral positive immune cells.
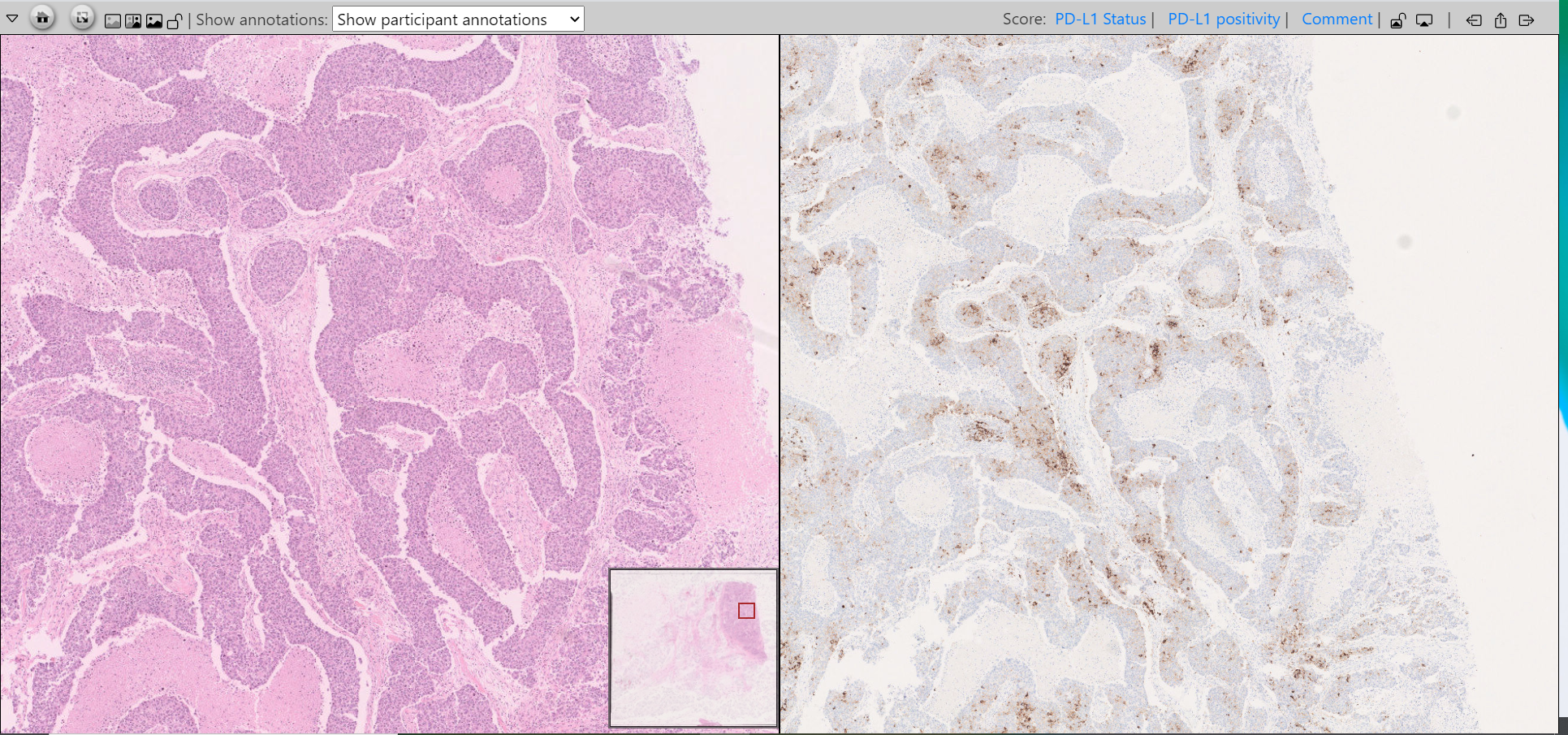
Screenshot from the UK NEQAS ICC & ISH PD-L1 module for TNBC
The module is currently in pilot, but UK NEQAS ICC & ISH plans to make it a regular module, offered 3-4 times a year, so keep an eye out for further announcements if you are interested in joining, or sign up for the PathoGate mailing list - and we will keep you posted.

